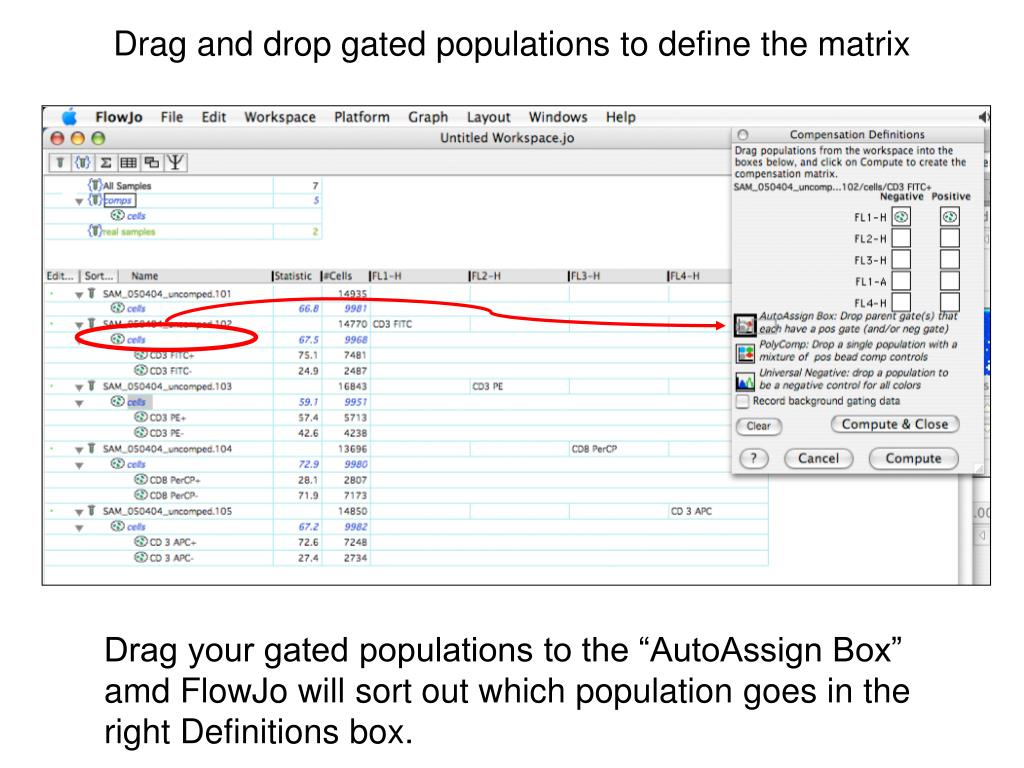
When making multiple matrices like this, you only need to repeat the drags of the successive Cy5PE populations followed by steps 4 and 5, saving a different matrix for each set of reagents. You will need to collect a singly-stained compensation control sample for each different Cy5PE reagent, and then construct a different matrix for each of these. Different Cy5PE reagents (even from the same manufacturer) have different spectra and thus require different compensations. This is true, for instance, when you are using different lots of tandem dyes (like Cy5PE). In some cases, you will need multiple compensation matrices for the same experiment. You can follow this example on your own computer by downloading the Compensation Demo Data. Display the compensated parameter (in brackets ) on the graph axes.Apply the compensation matrix to the appropriate samples.Drag each of the positive and negative populations from the workspace window into the appropriate box in the compensation dialog window.Bring up the Compensation Definition dialog window, or Compensation Wizard.Set a gate for the positive and negative populations for each singly-stained sample.We are indebted to our active and enthusiastic users worldwide for their ideas, discussions and tireless testing of new versions.ĭata Analysis Software for Flow CytometryAdvanced TutorialįlowJo was written by Adam Treister and Mario Roederer beginning in 1996, based on concepts developed at the Herzenberg laboratory at Stanford.This is a simple example showing how to do three-color compensation in FlowJo. FlowJo, its tutorials, documentation and web site are Copyright Tree Star, Inc. FlowJo Advanced Tutorial MMXRevision Date: FebruVersion 7.6įlowJo TutorialFlowJo is a software application designed to create an integrated environment for viewing and analyzing flow cytometric data. 
This environment is presented in the form of a Workspace. The Workspace contains a list of all of the data samples that you load, the gates, statistics and other analyses that you apply, and the table templates and graphical layout templates that you design. The Workspace is saved as a FlowJo document on your hard disk when you re-open the document, you will see the status of your analyses as they were when you last saved the Workspace. This tutorial is designed to introduce you to the program. Reading through it, you will learn how to operate FlowJo. Run the program as you perform the steps in the tutorial so that you can get the best feel for how the program works. As you watch FlowJo perform various operations such as creating new graphs, statistics, tables, or graphical layouts, you will see how fast and easy FlowJo is to use. It will take you three to six hours to complete the tutorial (you can easily break it up by chapter). The tutorial is designed around an example data set. The experiments used for the tutorial are based on 3-color immunophenotyping of human peripheral blood mononuclear cells (PBMC). The steps shown in this tutorial will help you in the analysis of nearly any kind of FACS data. The tutorial is only an introduction! FlowJo is capable of much that simply cant be covered in an introduction such as this (for example, there are Analysis Platforms to perform sophisticated DNA/Cell Cycle analysis, Kinetics analysis, Compensation, etc). You can learn more about FlowJo, and in particular how to use these platforms, through the on-line help facility. Whenever you ask for help from FlowJo, it launches a web browser and accesses help pages about the topic you selected. You can navigate the help pages to find out more about all aspects of FlowJo. The compensation control must be as least as bright as the stained sample. For the most accurate compensation, there are three basic rules that must be followed: 1. To access this on-line user manual directly, point your browser to The online help documentation provides tutorials for Compensation, Kinetics, and DNA/Cell Cycle analysis in addition, Tree Star provides Demonstration Data to let you explore these platforms. 3 Rules for Compensation Compensation uses single stained controls to account for fluorescence spillover and is critical for obtaining good multicolor flow cytometry data. CytoRSuite contains tools for every stage of the analysis pipeline including user-guided automatic compensation, realtime editing of spillover matrices, manual gate drawing, gate editing, visualisation of all existing flow cytometry objects and export of population level statistics for further analysis. In addition, there is a web page for FlowJo FAQs (Frequently-Asked Questions). An intuitive and interactive approach to flow cytometry analysis in R. #Flowjo 10 compensation tutorial manual.#Flowjo 10 compensation tutorial how to.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
